Speaking in scientific terms, electrochemistry is a branch of chemistry that studies the systems containing ions (charged particles), as well as processes and phenomena with their participation on the frontiers of phase interfaces (f.i., electrode and electrolyte). In other words, electrochemistry examines processes in which the oxidant and reductant are spatially separated, and the transfer of electrons is carried out by means of metal conductors.
What images should pop up in our mind when we hear this scientific definition? Svetlana Borisova, Coordinator of the Skoltech Center for Electrochemical Energy Storage, explains. Below please find excerpts from the interview with her.
Speaking about the practical application of electrochemistry, the first thing we have to mention is, of course, the creation and use of power sources: batteries, as well as fuel cells and electrolysis cells.
On the one hand, the lab is engaged with the basic research in electrochemistry, and on the other hand, is creating a more modern, cheaper energy storage devices with greater energy density. That is, with a larger amount of energy per unit volume or unit weight of the battery. The main task of the laboratory is to develop and test prototypes of batteries with improved properties.
The analytical part of the laboratory, which is already open and is actively functioning, is responsible for the study and analysis of the properties of various substances. That’s what we are going to talk about now.
What devices is the lab equipped with?
The first device the lab acquired is called a spectrophotometer for visible and ultraviolet areas of the spectrum. It helps us to carry out a spectral analysis of transparent environment. The device operates based on a simple physical principle meaning that different substances reflect and transmit light differently. For example, by illuminating the solution by a light of a certain wave length, we can define which materials it’s made of, as well as to determine the concentration of these substances.
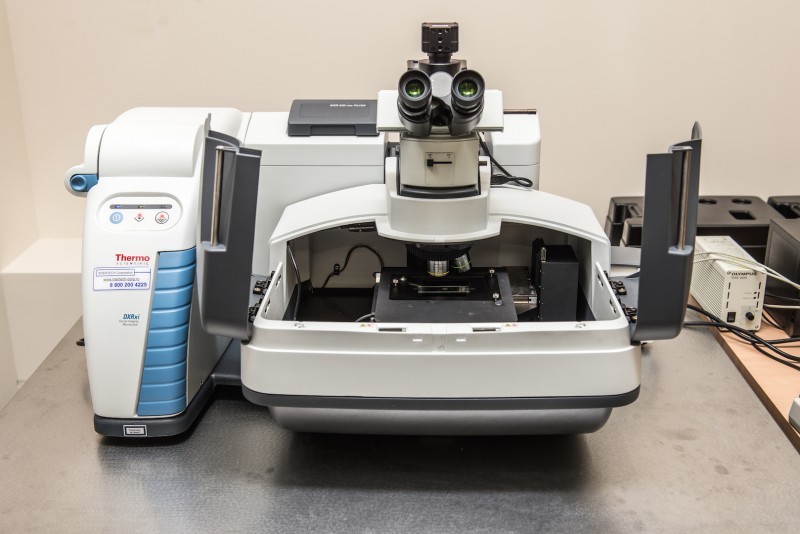
We have several modules for this device in our lab, which enable us to make measurements in different modes, depending on what task is set. For example, we can examine the fluid in a special cell for its transmission and light dissipation, to study thin films and to regulate the temperature of the samples. That is to figure out how the properties of various substances change at different temperatures. The computer that is connected to the device, records the detected spectra. And then we can compare them with the spectra of known substances to conclude what substance we deal with, and what its chemical composition is. This analysis makes no effect on the substance and, therefore, relates to methods for non-destructive analysis, as well as our next device does – The Thermo Scientific™ DXR™xi Raman Imaging Microscope produced by Thermo Fisher Scientific.
This device is the first one in Russia in this series. It allows for fast non-destructive analysis of materials, which also does not require any special sample preparation. In particular, this microscope allows to carry out a study of samples in a transparent envelope, as well as in solutions. With the new software the microscope enables to select imaging parameters of the received data in real time. The device operates as follows: we cover our samples with a laser beam, and a detector captures the spectrum of light which is inelastically dissipated on the molecules of this sample.
By to the obtained spectra we can make a conclusion on the composition and properties of the substance. The program also allows to compare the spectra obtained with the reference values for a large number of substances collected in digital libraries of the Raman spectra. In addition, the device makes it possible to quickly create entire maps for the samples, consisting of several substances. The microscope enables us to make measurements at both normal room temperature and at low temperatures. For this purpose we use a special add-on device for cooling the sample with liquid nitrogen.
Our laboratory already boasts electrochemical equipment: such as a potentiostat-galvanostat with a dedicated stand for electrochemical cells. This device allows us to study the characteristics of electrochemical cells and batteries. When we speak about the development of new materials for batteries, we are refeerring to the improvement of their properties and characteristics of the existing batteries.
A stand for electrochemical cells enables us to apply a variety of electrochemical methods for the characterization of electrode materials. This is done by assembling an electrochemical cell – a prototype battery. This allows to optimize the selection of anode and cathode materials and electrolytes.
Needless to say that when a new material for the battery is created, it needs to be tested. Then we connect the battery to the potentiostat, provide the constant voltage or current and register the curve of charge / discharge. The potentiostatic mode, respectively, allows us to study the reaction of batteries or electrochemical cells at a constant voltage, and galvanostatic mode – at constant current. Test results are the following characteristics of the studied battery, for example, we see how fast it is charged and discharged, and how many times we can repeat this procedure without the loss of battery capacity.
Can students and graduate students access the lab?
 As an example we set the European approach to working in the lab. All those MSc and PhD students working and studying in our Center for Electrochemical Energy Storage and who have been trained to work with devices, can independently carry out measurements in the laboratory. In addition, each device in our lab is matched with a PhD student who is responsible for it. This student is the contact person and consultant for other laboratories as well as for our students.
As an example we set the European approach to working in the lab. All those MSc and PhD students working and studying in our Center for Electrochemical Energy Storage and who have been trained to work with devices, can independently carry out measurements in the laboratory. In addition, each device in our lab is matched with a PhD student who is responsible for it. This student is the contact person and consultant for other laboratories as well as for our students.
As for the other Skoltech labs, we are ready to discuss the cooperation, and the joint research and scientific projects with the use of our devices. We are open to any partnerships – with both Skoltech partners, and any other interested research organizations and industry representatives.
What would be the final product of the lab?
Our laboratory is part of the Skoltech Center – or CREI – on electrochemical energy storage. In Russian the CREI abbreviation means “Center of science, innovation and education.” Accordingly, there will be three final products of our Center’s lab.
Speaking about science, it is, first and foremost, important scientific results, reflected in high-level publications, dissertations and diplomas of Skoltech PhD and MSc students. The main result of education is the preparation of qualified experts of the international level. Those would be students with high knowledge of the world science and industry, able to conduct independent research, able to work on the devices provided in the laboratory. Finally, innovation – it’s when the research results reach critical mass and get converted into commercial products, meeting the needs of industry.
In the middle of next year we are planning to open the second part of the lab, which will enhance our analytical capabilities, as well as will contain equipment for the synthesis of materials, assemblying and testing of prototypes of new types of batteries.
This final product will connect science, education and innovation!
Prepared by Anna Shimanskaya
Photo – Sk.ru