Researchers from Skoltech and their US colleagues have designed a new machine learning-based approach for detecting atrial fibrillation drivers, small patches of the heart muscle that are hypothesized to cause this most common type of cardiac arrhythmia. This approach may lead to more efficient targeted medical interventions to treat the condition that is estimated to affect more than 33 million people worldwide, according to the American Heart Association. The recent paper was published in the journal Circulation: Arrhythmia and Electrophysiology.
Researchers from Skoltech and their US colleagues have designed a new machine learning-based approach for detecting atrial fibrillation drivers, small patches of the heart muscle that are hypothesized to cause this most common type of cardiac arrhythmia. Credit: Pavel Odinev / Skoltech
The mechanism behind atrial fibrillation (AF), a type of abnormal heart rhythm that is associated with increased risk of heart failure and stroke, is yet unclear. Research suggests it may be caused and maintained by what’s called reentrant AF drivers, highly localized sources of repetitive rotational activity that lead to irregular heart rhythm. These drivers can be burnt via a surgical procedure, which can mitigate the condition or even restore the normal functioning of the heart.
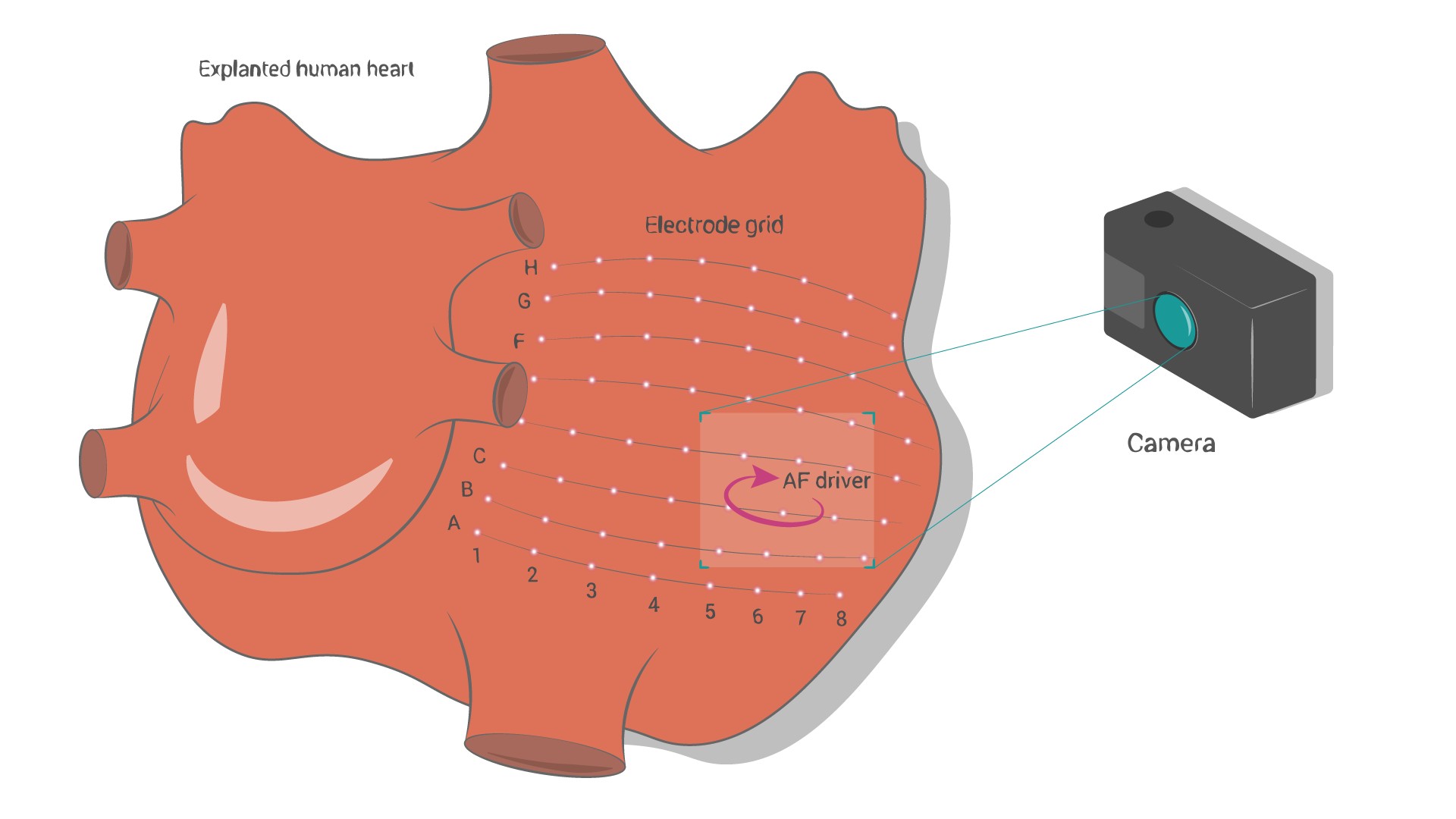
To locate these reentrant AF drivers for subsequent destruction, doctors use multi-electrode mapping, a technique that allows them to record multiple electrograms inside the heart (this is done with a catheter) and build a map of electrical activity within the atria. However, clinical applications of this technique often produce a lot of false negatives, when an existing AF driver is not found, and false positives, when a driver is detected where there really is none.
Recently, researchers have tapped machine learning algorithms for the task of interpreting ECGs to look for atrial fibrillation; however, these algorithms require labeled data with the true location of the driver, and the accuracy of multi-electrode mapping is insufficient. The authors of the new study, co-led by Dmitry Dylov from the Skoltech Center of Computational and Data-Intensive Science and Engineering (CDISE) and Vadim Fedorov from the Ohio State University, used high-resolution near-infrared optical mapping (NIOM) to locate AF drivers and stuck with it as a reference for training.
“NIOM is based on well-penetrating infrared optical signals and therefore can record the electrical activity from within the heart muscle, whereas conventional clinical electrodes can only measure the signals on the surface. Add to this trait the excellent optical resolution, and the optical mapping becomes a no-brainer modality if you want to visualize and understand the electrical signal propagation through the heart tissue,” says Dmitry Dylov.
The team tested their approach on eleven explanted human hearts, all donated posthumously for research purposes. Researchers performed the simultaneous optical and multi-electrode mapping of AF episodes induced in the hearts. ML model can indeed efficiently interpret electrograms from multielectrode mapping to locate AF drivers, with an accuracy of up to 81%. They believe that larger training datasets, validated by NIOM, can improve machine learning-based algorithms enough for them to become complementary tools in clinical practice.
“The dataset of recording from 11 human hearts is both extremely priceless and too small. We realized that clinical translation would require a much larger sample size for representative sampling, yet we had to make sure we extracted every piece of available information from the still-beating explanted human hearts. Dedication and scrutiny of two of our PhD students must be acknowledged here: Sasha Zolotarev spent several months on the academic mobility trip to Fedorov’s lab understanding the specifics of the imaging workflow and present the pilot study at the HRS conference – the biggest arrhythmology meeting in the world, and Katya Ivanova partook in the frequency and visualization analysis from within the walls of Skoltech. These two young researchers have squeezed out everything one possibly could, to train the machine learning model using optical measurements,” Dylov notes.
Other organizations involved in this research include The Ohio State University Wexner Medical Center and Davis Heart & Lung Research Institute. It was funded by grants from the NIH, the Russian Foundation for Basic Research, and the Bob and Corrine Frick Center for Heart Failure and Arrhythmia.
Contact information:
Skoltech Communications
+7 (495) 280 14 81
